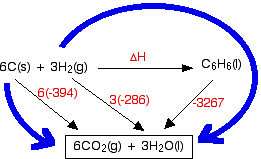
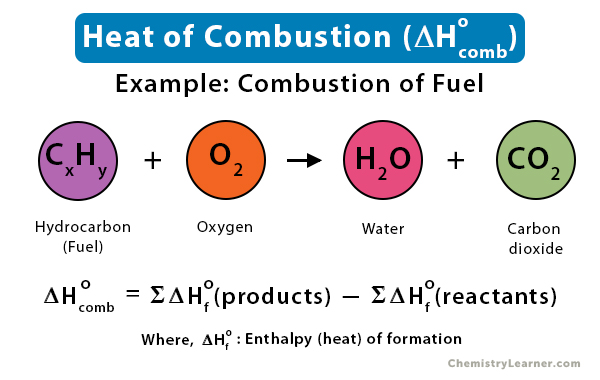
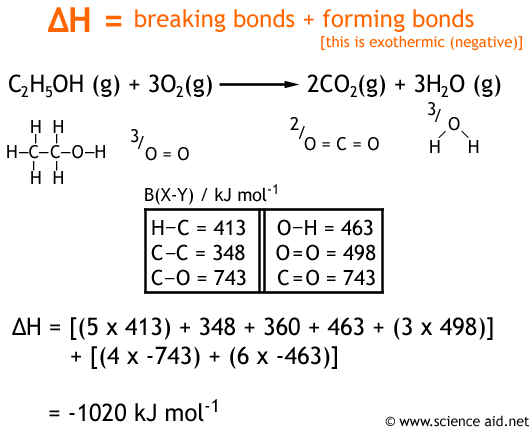
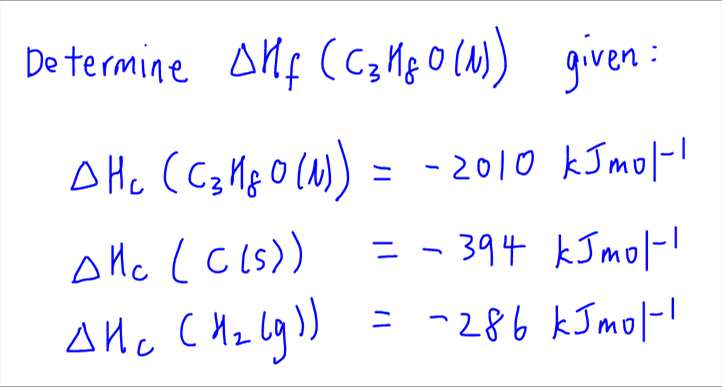Question Video: Determining the Standard Enthalpy of Formation of Ethanol Using Standard Enthalpies of Combustion | Nagwa

Calculate the enthalpy of combustion of ethylene at 1 atm pressure and 298 K, if enthalpy of formation of CO2(g) , H2O(I) and C2H4(g) are - 394, - 242 and - 52 kJ respectively.

Chemical Energetics: Experimental Method to Determine Enthalpy Change of Combustion - A-Level H2 Chemistry Tuition by 10 Year Series Author
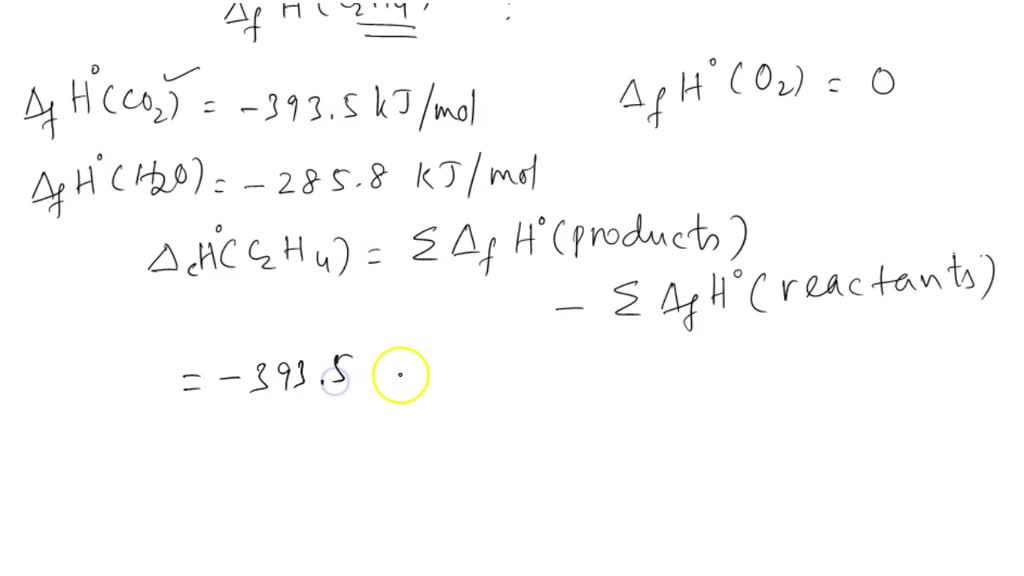
SOLVED: The standard enthalpy of combustion of ethene gas, C2H4 (g), is -1411.1 kJ/mol at 298 K. Given the following enthalpies of formation, calculate ΔHfo for C2H4 (g) CO2 (g): -393.5 kJ/mol

Question Video: Calculating Standard Enthalpy of Combustion of Methane Using Standard Enthalpies of Formation of Methane and Carbon Dioxide | Nagwa