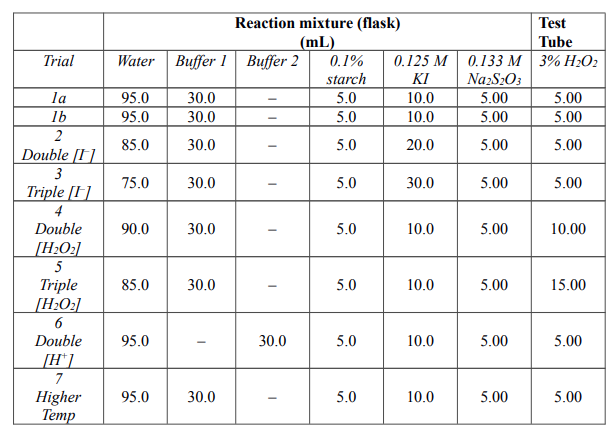
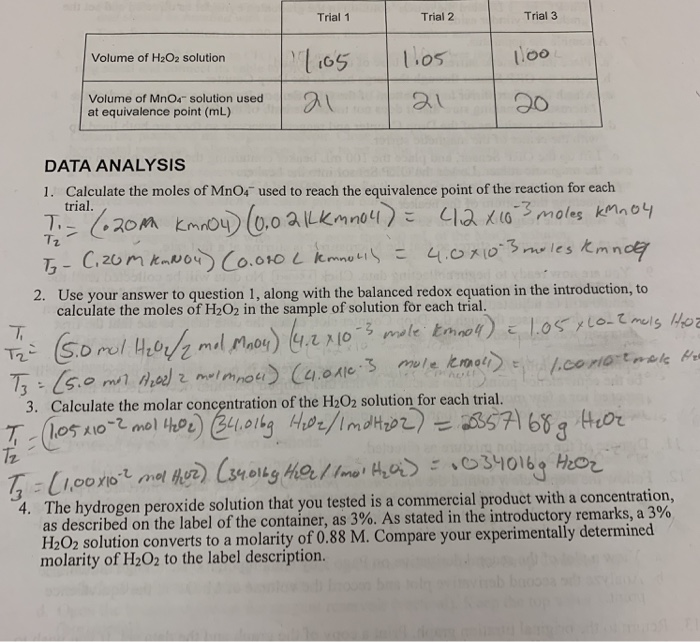
SOLVED: one method of determining the concentration of hydrogen peroxide ( H2O2) in a solution is through titration with the iodide ion. The net ionic equation is H2O2 + 2I-+2H+ –> I2+ 2H2O.
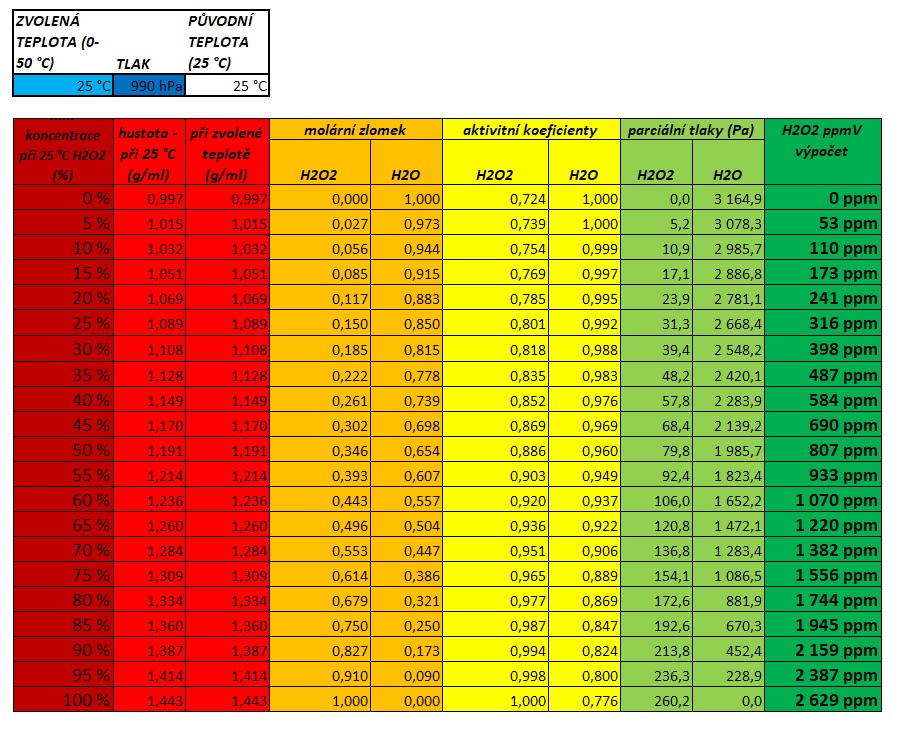
Calculator the equilibrium concentration of gaseous hydrogen peroxide over solution – Lukáš Kolík – physico-chemical calculation – working a personal webpage
Welcome to Chem Zipper.com......: Calculate the Molarity of H2O2 if 11.2 ml H2O2 require 30 ml of 0.5 M K2Cr2O7 for its Oxidation . also calculate the volume of strength of H2O2.


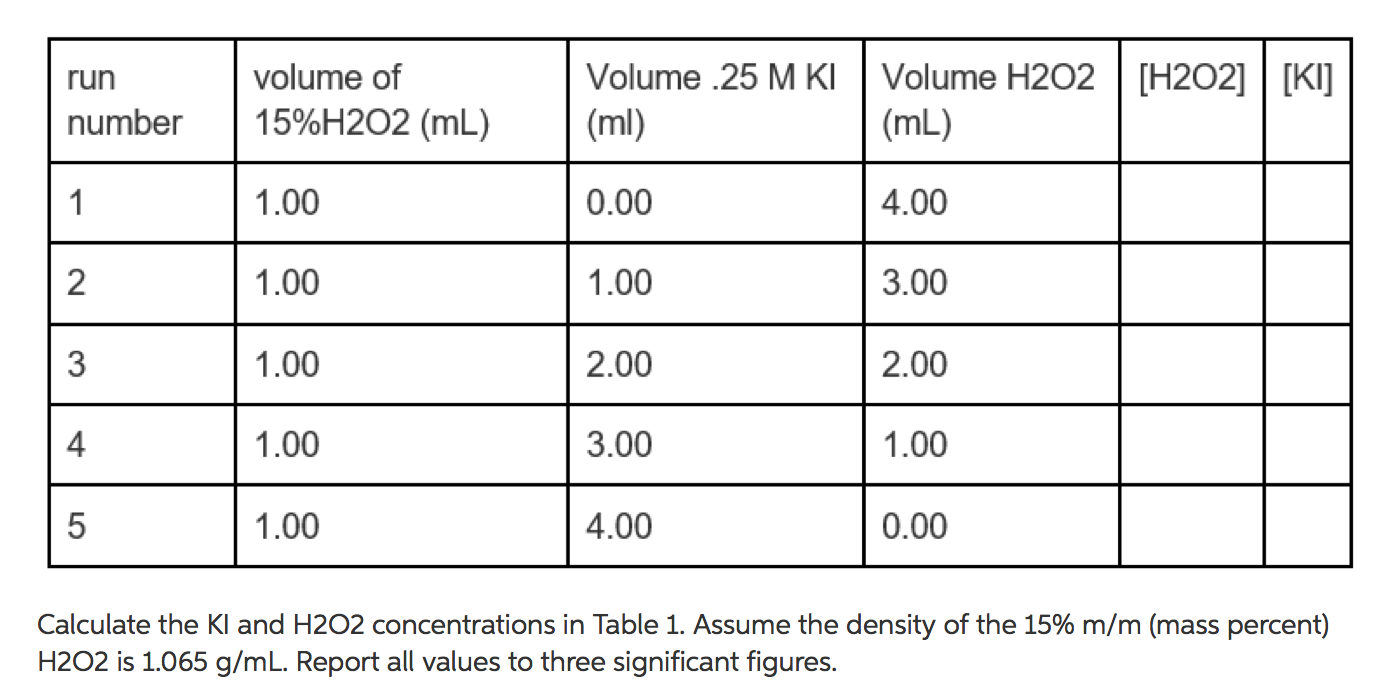


.gif)



![Hydrogen Peroxide [H2O2] Molecular Weight Calculation - Laboratory Notes Hydrogen Peroxide [H2O2] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2021/11/hydrogen-peroxide-molecular-weight-calculation.jpg)