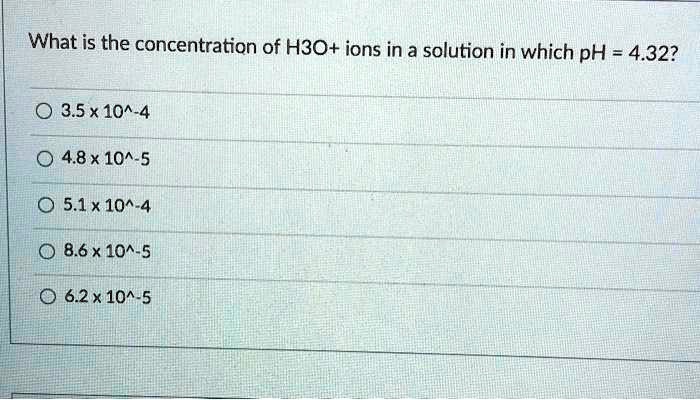
SOLVED: What is the concentration of H3O+ ions in a solution in which pH = 4.32? 3.5x 10^-4 4.8x 10^-5 0 5.1*10^-4 8.6 x 10^-5 6.2*10^-5
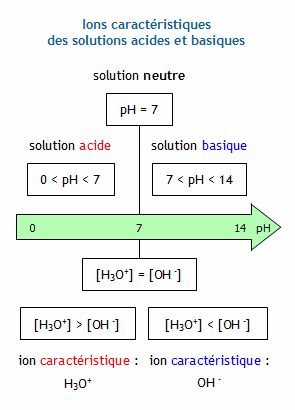
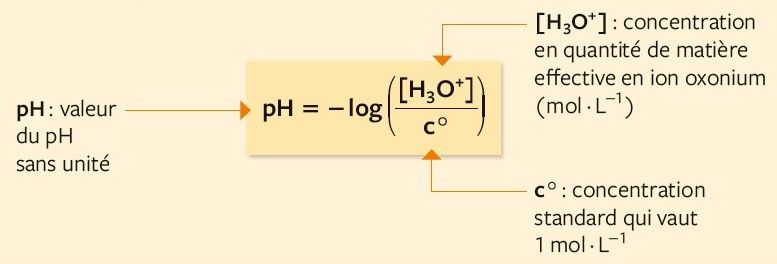
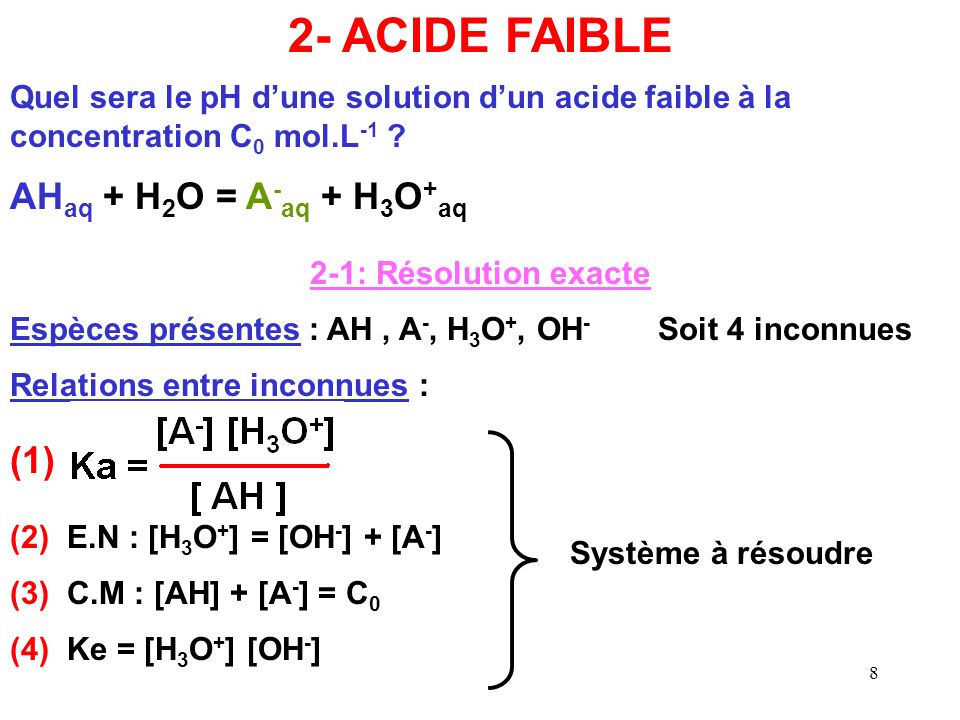
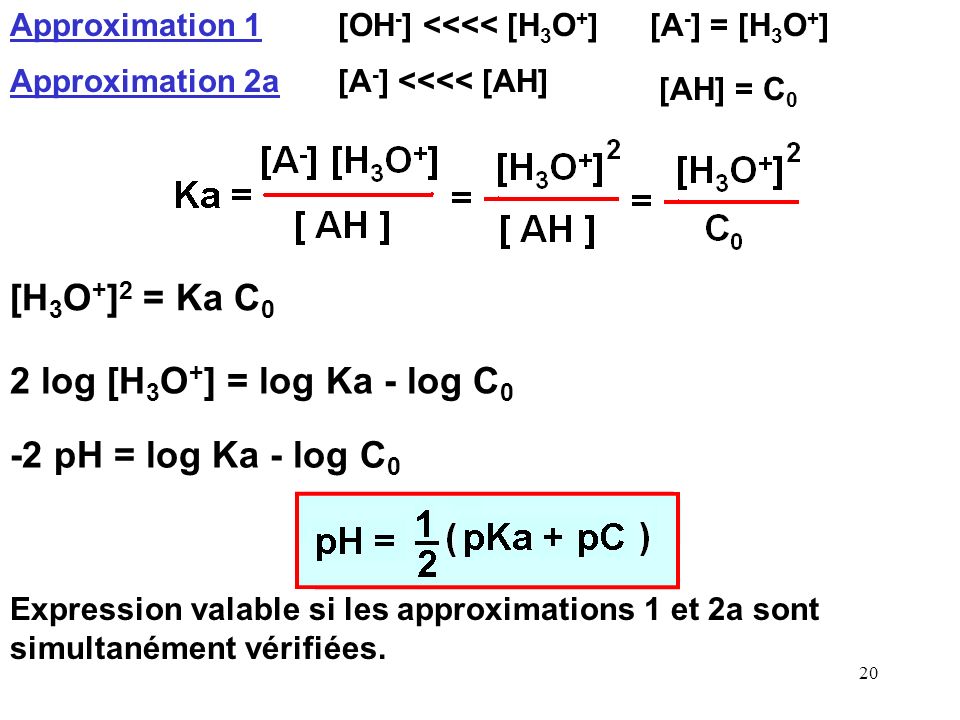
Calculer la concentration molaire en ions hydrogène et en ions hydroxyde d'une solution connaissant son pH

SOLVED: Calculating pH from hydrogen ion concentration and significant figure note: To calculate the pH from the hydrogen ion concentration; take the negative log of the molarity of hydrogen ion. This may
![SOLVED: What are the concentrations of H3O and OH– in oranges that have a pH of 3.98? [H3O+]= M [OH-]= M SOLVED: What are the concentrations of H3O and OH– in oranges that have a pH of 3.98? [H3O+]= M [OH-]= M](https://cdn.numerade.com/ask_previews/e9f7adf9-a742-4ec8-bdea-5a0f18192bbf_large.jpg)
SOLVED: What are the concentrations of H3O and OH– in oranges that have a pH of 3.98? [H3O+]= M [OH-]= M

SOLVED: 1. What is the pH of a urine sample that has an H3O+ concentration of 1 x 10-5 M? Classify the solution as acidic, basic or neutral. 2. What is the

Calculer le pH d'un acide fort & d'une base forte avec les approximations - E-Learning Chimie - YouTube

Vidéo de question : Identifier l'équation utilisée pour calculer la concentration en ions hydronium | Nagwa