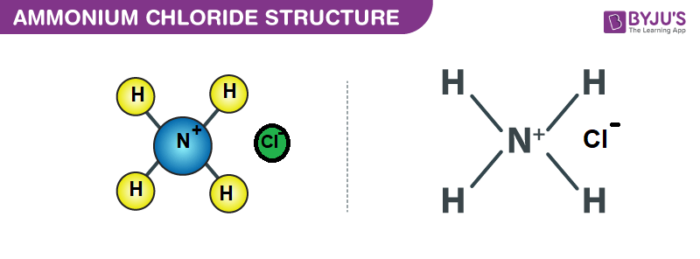
Ammonium Chloride (NH4Cl) - Structure, Properties, Preparation, Uses, Health Risk & FAQs of Ammonium Chloride.

Ammonium-chloride-assay - Assay Of Ammonium Chloride AIM: To determine the percentage purity of - Studocu

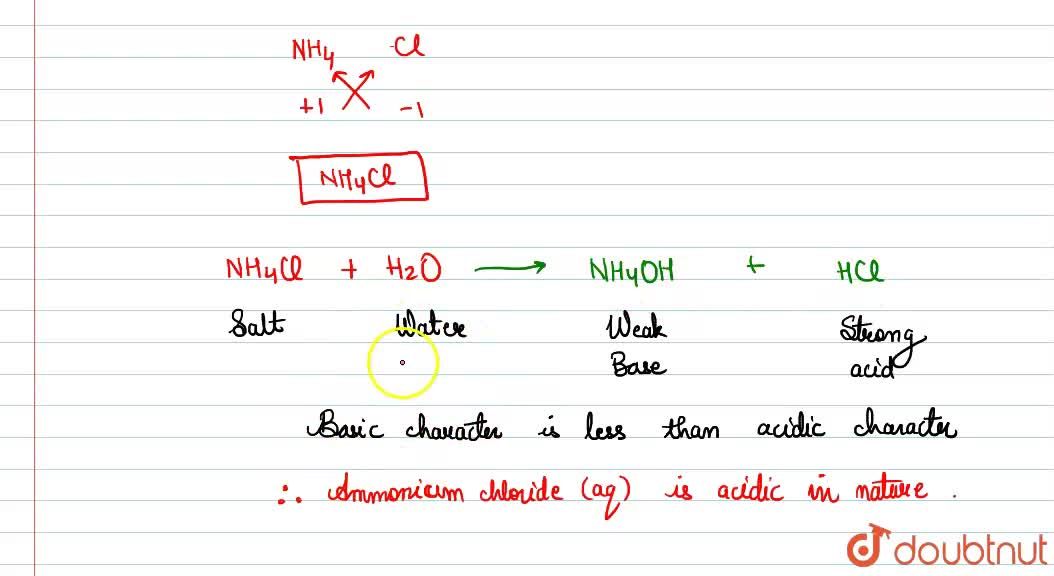
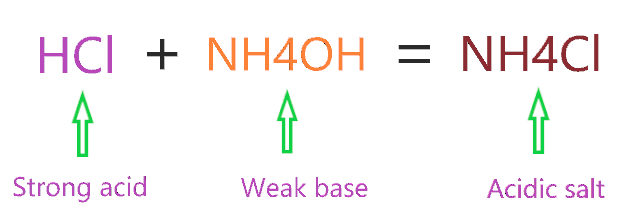
Identity the acid and base for ammonium chloride salt. state the nature of this salt and mention its - Brainly.in

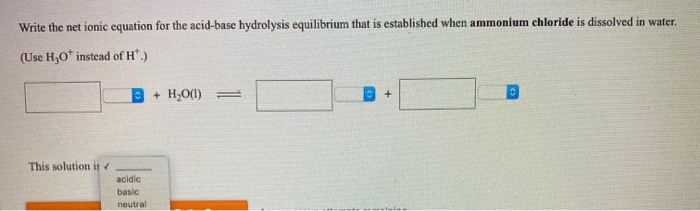
Write the chemical formula of ammonium chloride. Explain why an aueous solution of ammonium chloirde - YouTube

SOLVED: In a blood buffer, ammonium chloride (NH4Cl) was added to lower the blood pH. The ammonium ion is what acts as the acid. The chloride ion does not have any acid/base

pH calculation of a buffer solution made from a weak base and its conjugate acid (salt form) - YouTube













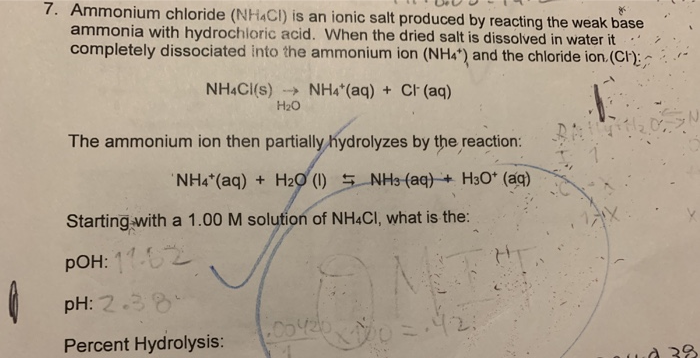
![Solved BUFFERS 10. An ammonium[NH3]/ammonium chloride | Chegg.com Solved BUFFERS 10. An ammonium[NH3]/ammonium chloride | Chegg.com](https://media.cheggcdn.com/media/98f/s907x655/98fc93a3-c6f6-4a89-8acb-45d7d9f5eb4d/image.png)