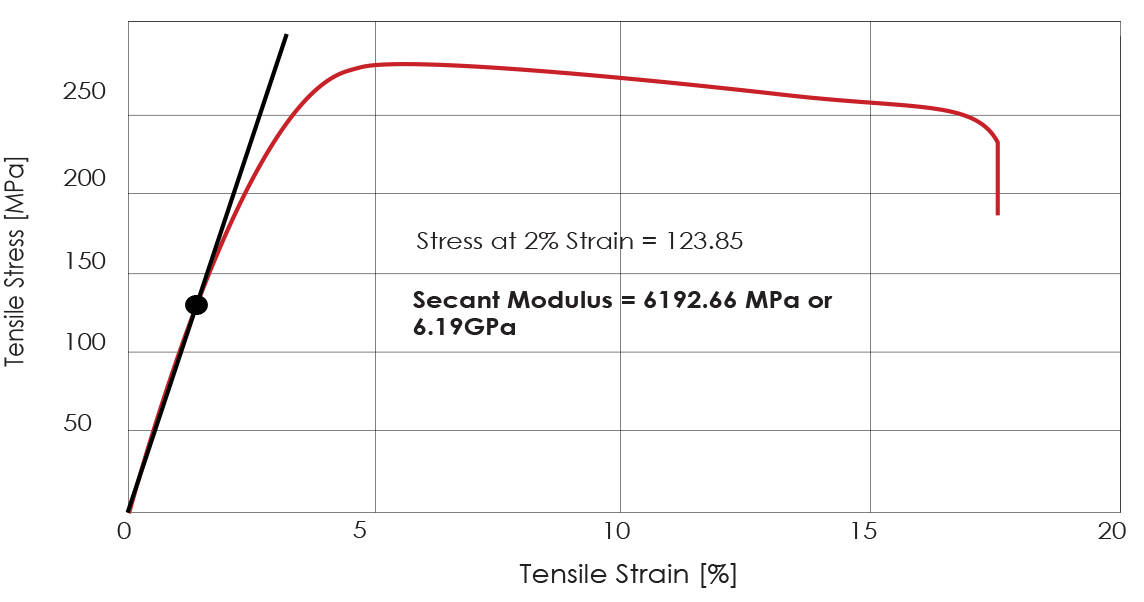
SOLVED:Use the Born-Haber cycle and data from Appendix IVB and Table 10.3 to calculate the lattice energy of to calculate the lattice energy of CaO. ΔHsub for calcium is 178 kJ /

Orientation Workbook - Orientation Module Self-assessment rubric Please indicate your selections - StuDocu