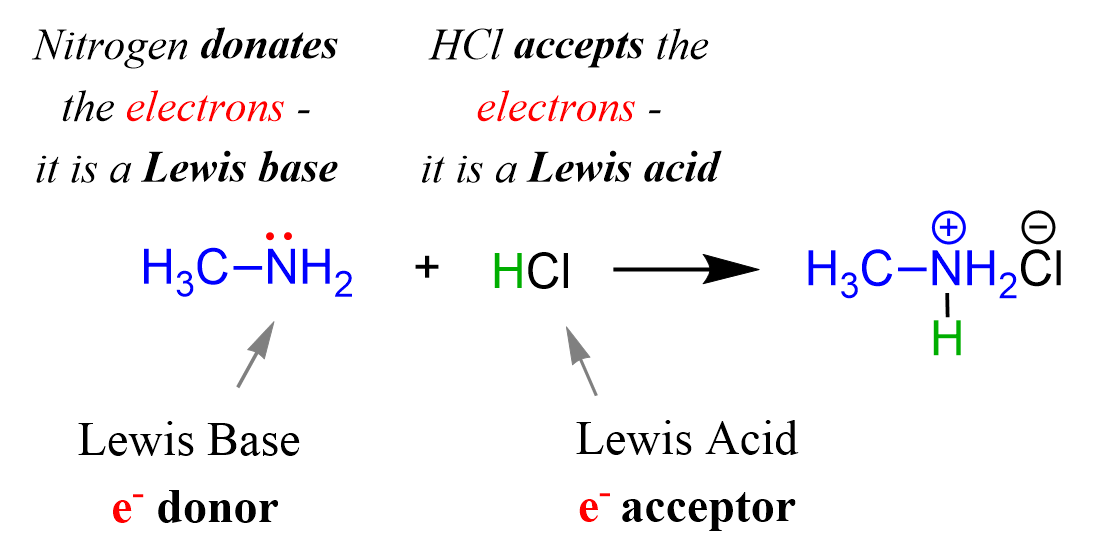
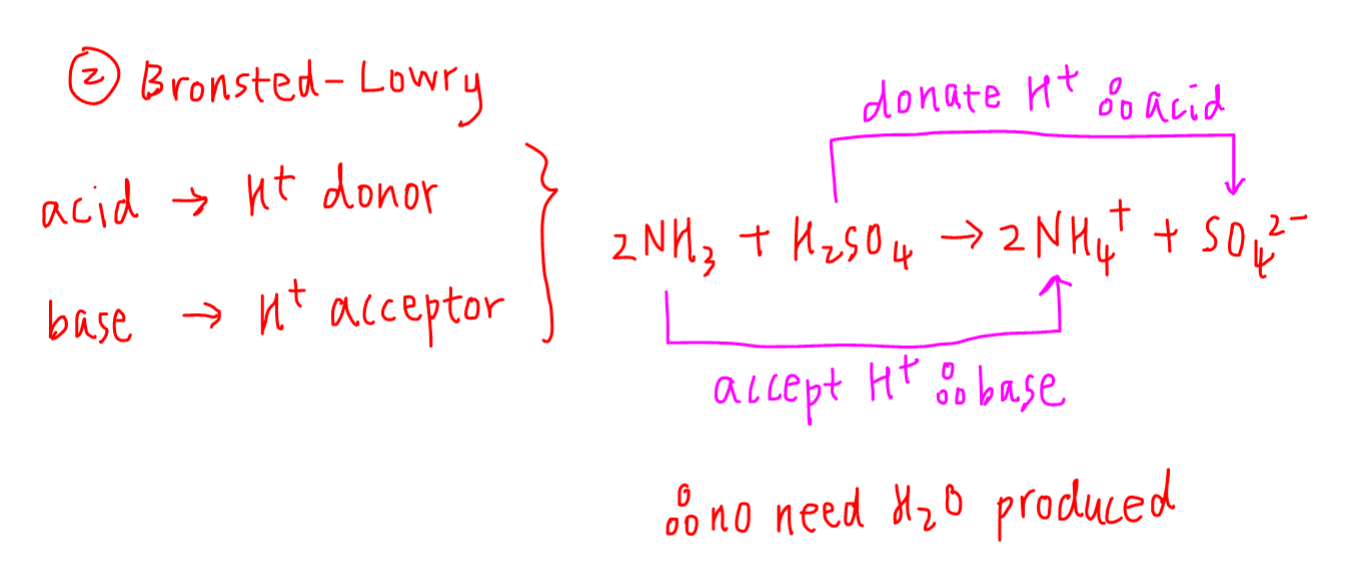
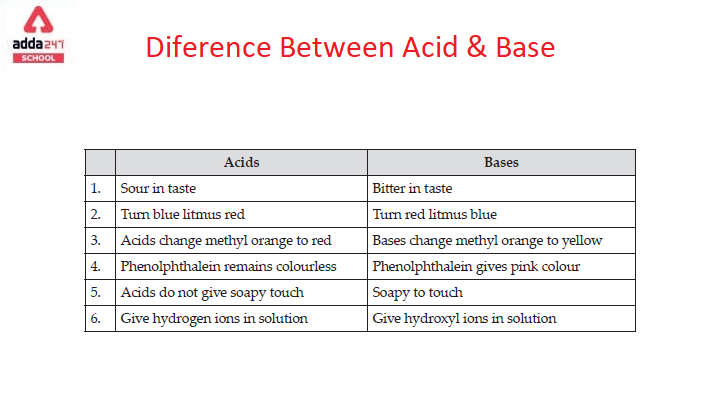
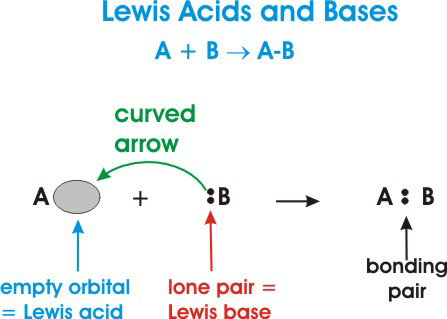
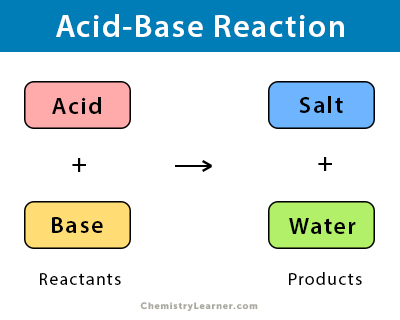
Acids and Bases 3 definitions for acids and bases – Arrhenius – Bronsted-Lowry – Lewis Must be in solution – Most often dissolved in water (aqueous) Inorganic. - ppt download
![Acids and Bases Topics to be covered: Definitions of acids and bases; Bronsted's conjugate acid-base pairs concept; Determination of [H 3 O + ], [OH - - ppt download Acids and Bases Topics to be covered: Definitions of acids and bases; Bronsted's conjugate acid-base pairs concept; Determination of [H 3 O + ], [OH - - ppt download](https://images.slideplayer.com/20/6056284/slides/slide_2.jpg)
Acids and Bases Topics to be covered: Definitions of acids and bases; Bronsted's conjugate acid-base pairs concept; Determination of [H 3 O + ], [OH - - ppt download

SOLVED: Determine if each statement below is Trueor Ealse regarding Arrhenius and Bronsted-Lowry definitions for acids and bases. 1) An Arrhenius acid is a substance that dissolves in water to produce Ht

Acids, Bases and Buffers The Br Ø nsted-Lowry definitions of an acid and a base are: Acid: species that donates a proton Base: species that can accept. - ppt download

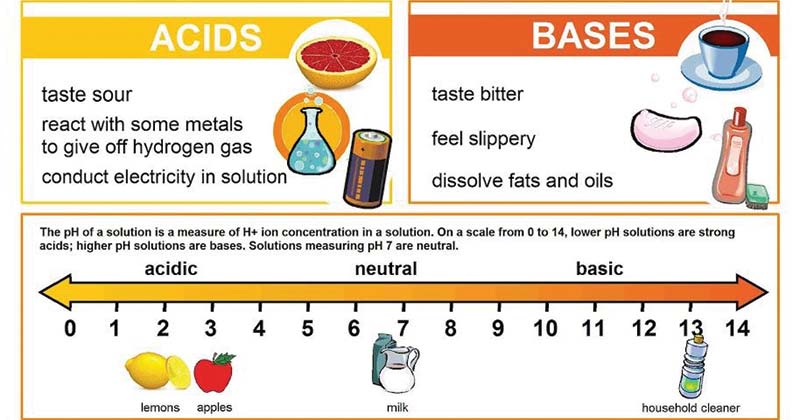
Definitions. Arrhenius Acids and Bases Acids release hydrogen ions in water. Bases release hydroxide ions in water. An acid is a substance that produces. - ppt download

SOLVED: TABLE 14.10 Three Models for Acids and Bases Model Arrhenius Bronsted Lowry Lewis Delinition of Acid producer Definition of Base OH producer acceptor Electron-pair donor donor Electron-pair acceptor



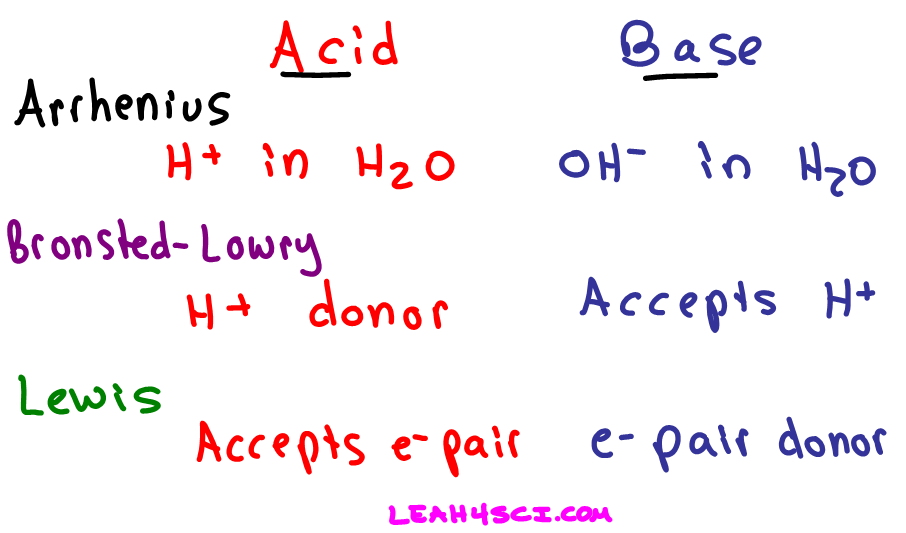



:max_bytes(150000):strip_icc()/one-ambidextrous-indian-man-writing-chemical-equation-on-greenboard-154948585-5880bed45f9b58bdb32f7efb.jpg)